Dec 16 , 2021. 11 hours ago – 23:14 KYODO NEWS
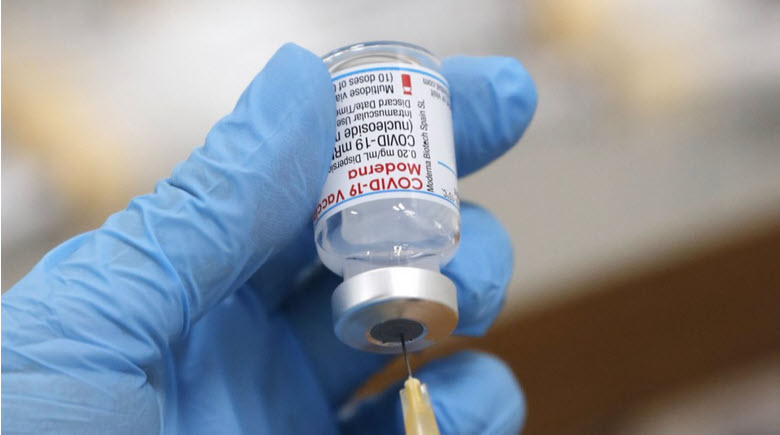
TOKYO – A Japanese health ministry panel approved Wednesday the administration of U.S. biotechnology firm Moderna Inc.’s COVID-19 booster shots at least six months after an individual aged 18 or older has received their second dose.
The third dose will use half the 0.5 milliliter amount used for each of the first two shots. Moderna’s COVID-19 vaccine is the second to be approved for use as booster shots in Japan after Pfizer Inc. received the green light last month.
Japan has set an eight-month interval “in principle” between second vaccination shots and a booster but has been working to bring forward the third shots following the emergence of the Omicron variant of the coronavirus.
But while numerous cases of the variant have been confirmed in Japan, daily infections nationwide have remained at under 200 for the past few weeks.
Takeda Pharmaceutical Co., which is in charge of sales and distribution of the Moderna vaccine in Japan, applied for approval last month to administer the third shot with a shortened interval of at least six months.
The Ministry of Health, Labor and Welfare will officially grant fast-track approval to the drugmaker’s vaccine for use as booster shots as early as Thursday.
The booster shots, which will be provided free of charge, are expected to be used in the rollout for the general public beginning in January, as well as for workplace vaccinations starting in March.
Japan is due to receive 50 million doses of Moderna next year. But as the dosage will be halved for booster shots, the supply will yield at least 1.5 times more shots at 75 million.
The government began administering third doses of Pfizer’s vaccine to health care workers earlier this month, with those for the elderly set to start in many municipalities in January.
It has said that a booster vaccine can be a different brand from the previous two shots administered, and Moderna’s can also be used for mixing and matching if approved.
In a separate development, the ministry will convene a special committee, possibly on Dec. 24, to decide whether to grant approval to molnupiravir, an oral COVID-19 drug developed by U.S. pharmaceutical giant Merck & Co., according to an official familiar with the matter.
The antiviral drug, which prevents the virus from entering or multiplying in the body, could be the first such pill to be used in Japan if approved.
The company’s Japanese arm MSD K.K. has said that molnupiravir is likely effective against the Omicron coronavirus variant.
The Japanese government has already agreed with the subsidiary to pay about $1.2 billion to procure molnupiravir doses for 1.6 million patients.
Early last month, Britain became the first country in the world to approve the use of the drug.
Cr.KYODO NEWS
